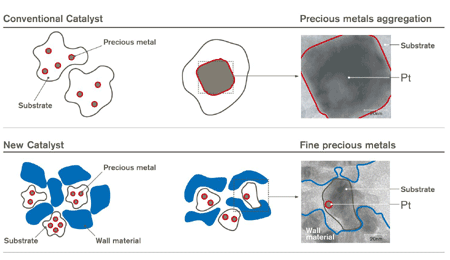
Nissan Motor Co., Ltd has developed a new catalyst for petrol engines that uses only half the precious metals of conventional catalysts.
A catalyst uses platinum, rhodium and palladium to interact with the exhaust gases causing chemical reactions with nitrogen oxide (NOX), carbon monoxide (CO) and hydrocarbons (HC) – into non-toxic compounds such as nitrogen (N2), water (H20), and carbon dioxide (CO2).
However, the high temperatures of the exhaust gas causes the precious metals to cluster up, reducing exposed metal surface area, thus reducing the effectiveness of the catalyst over time. So what manufacturers have done is use excessive amounts of precious metals so that the catalyst still functions sufficiently well even after the effectiveness has been reduced.
Nissan has used nano-technology to keep the fine metal particles separated under high temperatures, so that only the necessary amount of precious metals need to be used – about 50% less. This will maintain cleaner emissions as well as reduce the consumption of precious metals – an effort of Nissan’s Green Program 2010.
Looking to sell your car? Sell it with Carro.

















greener is always s great thing…… ;)
hmm…
nice idea, not sure how they will use it in their ad campaign to sell more cars